for an atom to achieve maximum stability and become chemically inert, what must occur?
Overview of Atomic Structure
Atoms are fabricated up of particles called protons, neutrons, and electrons, which are responsible for the mass and charge of atoms.
Learning Objectives
Discuss the electronic and structural properties of an cantlet
Cardinal Takeaways
Key Points
- An atom is composed of two regions: the nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus.
- Protons and neutrons have approximately the same mass, near i.67 × 10-24 grams, which scientists define every bit ane diminutive mass unit of measurement (amu) or 1 Dalton.
- Each electron has a negative charge (-one) equal to the positive charge of a proton (+i).
- Neutrons are uncharged particles found within the nucleus.
Key Terms
- cantlet: The smallest possible corporeality of matter which still retains its identity equally a chemical element, consisting of a nucleus surrounded by electrons.
- proton: Positively charged subatomic particle forming role of the nucleus of an atom and determining the atomic number of an element. It weighs one amu.
- neutron: A subatomic particle forming role of the nucleus of an atom. It has no accuse. Information technology is equal in mass to a proton or it weighs i amu.
An atom is the smallest unit of measurement of matter that retains all of the chemical backdrop of an chemical element. Atoms combine to course molecules, which then interact to form solids, gases, or liquids. For example, water is composed of hydrogen and oxygen atoms that have combined to course h2o molecules. Many biological processes are devoted to breaking downward molecules into their component atoms so they tin can be reassembled into a more useful molecule.
Atomic Particles
Atoms consist of three basic particles: protons, electrons, and neutrons. The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). The outermost regions of the atom are called electron shells and incorporate the electrons (negatively charged). Atoms accept different properties based on the arrangement and number of their bones particles.
The hydrogen cantlet (H) contains only one proton, one electron, and no neutrons. This can be determined using the atomic number and the mass number of the element (meet the concept on atomic numbers and mass numbers).
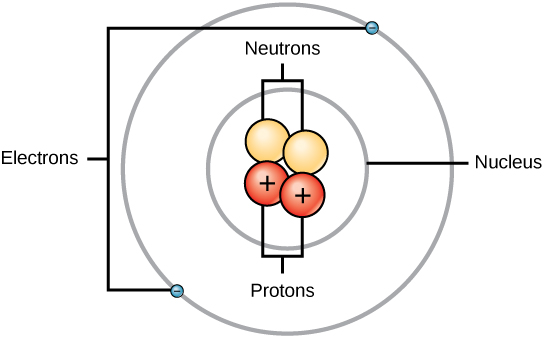
Structure of an atom: Elements, such every bit helium, depicted hither, are made up of atoms. Atoms are fabricated up of protons and neutrons located inside the nucleus, with electrons in orbitals surrounding the nucleus.
Atomic Mass
Protons and neutrons take approximately the same mass, about 1.67 × 10-24 grams. Scientists define this corporeality of mass as one atomic mass unit (amu) or i Dalton. Although similar in mass, protons are positively charged, while neutrons take no charge. Therefore, the number of neutrons in an atom contributes significantly to its mass, simply not to its charge.
Electrons are much smaller in mass than protons, weighing only 9.11 × 10-28 grams, or almost 1/1800 of an atomic mass unit. Therefore, they do not contribute much to an element'due south overall atomic mass. When considering atomic mass, it is customary to ignore the mass of any electrons and calculate the atom'southward mass based on the number of protons and neutrons alone.
Electrons contribute profoundly to the atom's charge, as each electron has a negative accuse equal to the positive charge of a proton. Scientists define these charges as "+1" and "-1. " In an uncharged, neutral atom, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus. In these atoms, the positive and negative charges cancel each other out, leading to an cantlet with no net charge.

Protons, neutrons, and electrons: Both protons and neutrons accept a mass of 1 amu and are found in the nucleus. However, protons have a charge of +1, and neutrons are uncharged. Electrons have a mass of approximately 0 amu, orbit the nucleus, and have a charge of -i.
Volume of Atoms
Accounting for the sizes of protons, neutrons, and electrons, most of the volume of an atom—greater than 99 percentage—is, in fact, empty space. Despite all this empty infinite, solid objects do non just pass through one some other. The electrons that surround all atoms are negatively charged and cause atoms to repel one another, preventing atoms from occupying the aforementioned space. These intermolecular forces prevent you from falling through an object similar your chair.
Atomic Number and Mass Number
The atomic number is the number of protons in an element, while the mass number is the number of protons plus the number of neutrons.
Learning Objectives
Determine the relationship between the mass number of an atom, its diminutive number, its atomic mass, and its number of subatomic particles
Key Takeaways
Key Points
- Neutral atoms of each chemical element incorporate an equal number of protons and electrons.
- The number of protons determines an element's atomic number and is used to distinguish 1 element from another.
- The number of neutrons is variable, resulting in isotopes, which are different forms of the aforementioned atom that vary only in the number of neutrons they possess.
- Together, the number of protons and the number of neutrons determine an chemical element's mass number.
- Since an element's isotopes have slightly dissimilar mass numbers, the atomic mass is calculated past obtaining the mean of the mass numbers for its isotopes.
Key Terms
- mass number: The sum of the number of protons and the number of neutrons in an atom.
- atomic number: The number of protons in an cantlet.
- atomic mass: The average mass of an cantlet, taking into account all its naturally occurring isotopes.
Atomic Number
Neutral atoms of an element comprise an equal number of protons and electrons. The number of protons determines an element's diminutive number (Z) and distinguishes one element from another. For case, carbon's atomic number (Z) is 6 because it has six protons. The number of neutrons can vary to produce isotopes, which are atoms of the same element that have unlike numbers of neutrons. The number of electrons can likewise be unlike in atoms of the same element, thus producing ions (charged atoms). For example, iron, Iron, tin can exist in its neutral state, or in the +ii and +three ionic states.
Mass Number
An element's mass number (A) is the sum of the number of protons and the number of neutrons. The small-scale contribution of mass from electrons is disregarded in calculating the mass number. This approximation of mass can exist used to hands calculate how many neutrons an element has by simply subtracting the number of protons from the mass number. Protons and neutrons both counterbalance about one atomic mass unit of measurement or amu. Isotopes of the aforementioned chemical element will have the same diminutive number but different mass numbers.

Atomic number, chemical symbol, and mass number: Carbon has an atomic number of half-dozen, and two stable isotopes with mass numbers of twelve and thirteen, respectively. Its average atomic mass is 12.11.
Scientists make up one's mind the atomic mass by calculating the hateful of the mass numbers for its naturally-occurring isotopes. Oftentimes, the resulting number contains a decimal. For example, the atomic mass of chlorine (Cl) is 35.45 amu considering chlorine is composed of several isotopes, some (the majority) with an atomic mass of 35 amu (17 protons and 18 neutrons) and some with an atomic mass of 37 amu (17 protons and twenty neutrons).
Given an atomic number (Z) and mass number (A), you can detect the number of protons, neutrons, and electrons in a neutral cantlet. For case, a lithium cantlet (Z=3, A=7 amu) contains three protons (found from Z), 3 electrons (as the number of protons is equal to the number of electrons in an atom), and four neutrons (7 – 3 = 4).
Isotopes
Isotopes are various forms of an element that have the same number of protons, but a unlike number of neutrons.
Learning Objectives
Talk over the properties of isotopes and their use in radiometric dating
Central Takeaways
Primal Points
- Isotopes are atoms of the aforementioned chemical element that contain an identical number of protons, but a different number of neutrons.
- Despite having dissimilar numbers of neutrons, isotopes of the same chemical element accept very similar physical properties.
- Some isotopes are unstable and volition undergo radioactive decay to become other elements.
- The predictable half-life of different decaying isotopes allows scientists to date material based on its isotopic composition, such as with Carbon-14 dating.
Key Terms
- isotope: Any of two or more forms of an chemical element where the atoms have the same number of protons, but a unlike number of neutrons within their nuclei.
- half-life: The time it takes for one-half of the original concentration of an isotope to decay back to its more stable form.
- radioactive isotopes: an atom with an unstable nucleus, characterized by excess energy bachelor that undergoes radioactive decay and creates most unremarkably gamma rays, blastoff or beta particles.
- radiocarbon dating: Determining the age of an object past comparing the ratio of the 14C concentration found in it to the corporeality of 14C in the atmosphere.
What is an Isotope?
Isotopes are diverse forms of an element that have the same number of protons just a different number of neutrons. Some elements, such equally carbon, potassium, and uranium, take multiple naturally-occurring isotopes. Isotopes are defined outset by their element and then by the sum of the protons and neutrons nowadays.
- Carbon-12 (or 12C) contains vi protons, six neutrons, and vi electrons; therefore, it has a mass number of 12 amu (half-dozen protons and six neutrons).
- Carbon-xiv (or fourteenC) contains six protons, eight neutrons, and six electrons; its atomic mass is 14 amu (six protons and viii neutrons).
While the mass of individual isotopes is different, their physical and chemical properties remain mostly unchanged.
Isotopes do differ in their stability. Carbon-12 (12C) is the most abundant of the carbon isotopes, bookkeeping for 98.89% of carbon on Globe. Carbon-14 (14C) is unstable and only occurs in trace amounts. Unstable isotopes well-nigh commonly emit blastoff particles (He2+) and electrons. Neutrons, protons, and positrons tin can likewise be emitted and electrons tin exist captured to attain a more than stable atomic configuration (lower level of potential energy ) through a process called radioactive decay. The new atoms created may be in a high free energy state and emit gamma rays which lowers the free energy but alone does non change the atom into another isotope. These atoms are called radioactive isotopes or radioisotopes.
Radiocarbon Dating
Carbon is normally nowadays in the atmosphere in the grade of gaseous compounds like carbon dioxide and marsh gas. Carbon-14 (14C) is a naturally-occurring radioisotope that is created from atmospheric xivN (nitrogen) by the addition of a neutron and the loss of a proton, which is caused past cosmic rays. This is a continuous process so more fourteenC is e'er being created in the temper. One time produced, the xivC oft combines with the oxygen in the atmosphere to course carbon dioxide. Carbon dioxide produced in this mode diffuses in the atmosphere, is dissolved in the ocean, and is incorporated by plants via photosynthesis. Animals eat the plants and, ultimately, the radiocarbon is distributed throughout the biosphere.
In living organisms, the relative corporeality of 14C in their body is approximately equal to the concentration of 14C in the atmosphere. When an organism dies, it is no longer ingesting xivC, so the ratio between 14C and 12C will decline as 14C gradually decays back to fourteenDue north. This ho-hum process, which is chosen beta decay, releases energy through the emission of electrons from the nucleus or positrons.
Later on approximately 5,730 years, one-half of the starting concentration of fourteenC will have been converted back to 14N. This is referred to as its half-life, or the time it takes for one-half of the original concentration of an isotope to decay back to its more stable form. Considering the half-life of fourteenC is long, information technology is used to date formerly-living objects such as onetime bones or wood. Comparing the ratio of the 14C concentration found in an object to the corporeality of xivC in the atmosphere, the corporeality of the isotope that has non notwithstanding decayed tin can be determined. On the basis of this amount, the age of the fabric can be accurately calculated, every bit long as the material is believed to exist less than 50,000 years old. This technique is called radiocarbon dating, or carbon dating for brusk.

Application of carbon dating: The age of carbon-containing remains less than 50,000 years onetime, such as this pygmy mammoth, can exist determined using carbon dating.
Other elements have isotopes with different half lives. For example, 40Grand (potassium-xl) has a one-half-life of 1.25 billion years, and 235U (uranium-235) has a half-life of well-nigh 700 million years. Scientists ofttimes apply these other radioactive elements to appointment objects that are older than l,000 years (the limit of carbon dating). Through the apply of radiometric dating, scientists can study the age of fossils or other remains of extinct organisms.
The Periodic Table
Everything in the universe is made of one or more than elements. The periodic tabular array is a ways of organizing the various elements according to similar physical and chemical properties.
Learning Objectives
Discuss the organization of the periodic table
Fundamental Takeaways
Key Points
- All thing is made from atoms of i or more than elements. Living creatures consist mainly of carbon, hydrogen, oxygen, and nitrogen (CHON).
- Combining elements creates compounds that may take emergent backdrop.
- The periodic tabular array is a list of the elements according to increasing atomic number that is farther organized into columns based on similar concrete and chemical properties and electron configuration.
- As i moves downward a column or beyond a row, there are some general trends for the properties of the elements.
- The periodic tabular array continues to expand today as heavier and heavier elements are created in laboratories around the globe.
Fundamental Terms
- element: Pure chemical substances consisting of only ane type of cantlet with a divers set up of chemical and physical backdrop.
- emergent properties: Properties establish in compound structures that are different from those of the individual components and would not be predicted based on the properties of the individual components.
- periodic table: A tabular chart of the chemical elements according to their atomic numbers and so that elements with like properties are in the same column.
Affair and Elements
Matter comprises all of the physical objects in the universe, those that take upwardly space and have mass. All affair is composed of atoms of one or more elements, pure substances with specific chemical and concrete properties. There are 98 elements that naturally occur on earth, yet living systems employ a relatively small number of these. Living creatures are composed mainly of just four elements: carbon, hydrogen, oxygen, and nitrogen (frequently remembered by the acronym CHON). As elements are bonded together they form compounds that often accept new emergent properties that are unlike from the backdrop of the individual elements. Life is an instance of an emergent property that arises from the specific collection of molecules found in cells.

Elements of the human body arranged by percent of full mass: There are 25 elements believed to play an active part in human health. Carbon, hydrogen, oxygen, and nitrogen make up approximately 96% of the mass in a man body.
The Periodic Tabular array
The different elements are organized and displayed in the periodic table. Devised by Russian chemist Dmitri Mendeleev (1834–1907) in 1869, the table groups elements that, although unique, share certain chemic properties with other elements. In the periodic table the elements are organized and displayed co-ordinate to their atomic number and are arranged in a serial of rows (periods) and columns (groups) based on shared chemic and concrete properties. If you look at a periodic table, you will see the groups numbered at the elevation of each column from left to right starting with 1 and ending with eighteen. In improver to providing the diminutive number for each chemical element, the periodic table too displays the element'south atomic mass. Looking at carbon, for example, its symbol (C) and name announced, as well as its diminutive number of six (in the upper left-hand corner) and its diminutive mass of 12.eleven.

The periodic table: The periodic table shows the atomic mass and diminutive number of each element. The atomic number appears above the symbol for the element and the approximate diminutive mass appears below it.
The organization of the periodic table allows the elements to be grouped according to their chemic backdrop. Inside the main group elements ( Groups 1-2, thirteen-18), in that location are some general trends that we can detect. The further down a given group, the elements have an increased metallic grapheme: they are skilful conductors of both oestrus and electricity, solids at room temperature, and shiny in advent. Moving from left to correct across a period, the elements have greater non-metallic character. These elements are insulators, poor oestrus conductors, and tin be in unlike phases at room temperature (brittle solid, liquid, or gas). The elements at the boundary between the metallic elements (grayness elements) and nonmetal elements (dark-green elements) are metalloid in grapheme (pink elements). They have low conductivity that increases with temperature. They also share properties with both the metals and the nonmetals.

The primary grouping elements: Inside the p-cake at the purlieus betwixt the metallic elements (grey elements) and nonmetal elements (green elements) at that place is positioned boron and silicon that are metalloid in character (pink elements), i.e., they take low electrical conductivity that increases with temperature.
Today, the periodic table continues to expand as heavier and heavier elements are synthesized in laboratories. These large elements are extremely unstable and, equally such, are very difficult to detect; just their continued creation is an ongoing challenge undertaken by scientists around the world.
Electron Shells and the Bohr Model
Niels Bohr proposed an early model of the atom as a central nucleus containing protons and neutrons being orbited by electrons in shells.
Learning Objectives
Construct an atom according to the Bohr model
Key Takeaways
Key Points
- In the Bohr model of the atom, the nucleus contains the majority of the mass of the atom in its protons and neutrons.
- Orbiting the positively-charged core are the negatively charged electrons, which contribute little in terms of mass, just are electrically equivalent to the protons in the nucleus.
- In nearly cases, electrons fill the lower- free energy orbitals commencement, followed by the next higher free energy orbital until information technology is full, so on until all electrons have been placed.
- Atoms tend to be well-nigh stable with a full outer trounce (one which, after the first, contains viii electrons), leading to what is usually called the " octet rule ".
- The properties of an element are determined by its outermost electrons, or those in the highest energy orbital.
- Atoms that practise non have full outer shells volition tend to gain or lose electrons, resulting in a full outer shell and, therefore, stability.
Cardinal Terms
- octet rule: A rule stating that atoms lose, gain, or share electrons in guild to have a total valence crush of 8 electrons. (Hydrogen is excluded because it can concord a maximum of 2 electrons in its valence shell. )
- electron shell: The collective states of all electrons in an atom having the same primary quantum number (visualized as an orbit in which the electrons motility).
Electron Shells and the Bohr Model

Orbitals in the Bohr model: The Bohr model was developed by Niels Bohr in 1913. In this model, electrons be inside principal shells. An electron usually exists in the lowest energy shell available, which is the 1 closest to the nucleus. Energy from a photon of calorie-free can bump information technology up to a higher energy shell, but this situation is unstable and the electron quickly decays back to the basis country. In the process, a photon of light is released.
As previously discussed, at that place is a connection betwixt the number of protons in an element, the atomic number that distinguishes one chemical element from another, and the number of electrons it has. In all electrically-neutral atoms, the number of electrons is the same as the number of protons. Each element, when electrically neutral, has a number of electrons equal to its atomic number.
An early model of the atom was developed in 1913 past Danish scientist Niels Bohr (1885–1962). The Bohr model shows the atom as a fundamental nucleus containing protons and neutrons with the electrons in circular orbitals at specific distances from the nucleus. These orbits class electron shells or energy levels, which are a way of visualizing the number of electrons in the various shells. These energy levels are designated by a number and the symbol "n." For example, 1n represents the first free energy level located closest to the nucleus.
Electrons make full orbit shells in a consequent guild. Nether standard weather condition, atoms fill the inner shells (closer to the nucleus) first, oft resulting in a variable number of electrons in the outermost beat. The innermost shell has a maximum of 2 electrons, just the next 2 electron shells can each take a maximum of 8 electrons. This is known as the octet rule which states that, with the exception of the innermost crush, atoms are more stable energetically when they take eight electrons in their valence trounce, the outermost electron shell. Examples of some neutral atoms and their electron configurations are shown in. Every bit shown, helium has a complete outer electron shell, with two electrons filling its first and only shell. Similarly, neon has a consummate outer 2n shell containing eight electrons. In contrast, chlorine and sodium take vii and one electrons in their outer shells, respectively. Theoretically, they would be more energetically stable if they followed the octet rule and had eight.

Bohr diagrams: Bohr diagrams betoken how many electrons make full each principal shell. Group xviii elements (helium, neon, and argon are shown) have a full outer, or valence, crush. A full valence shell is the most stable electron configuration. Elements in other groups have partially-filled valence shells and gain or lose electrons to achieve a stable electron configuration.
An cantlet may gain or lose electrons to attain a full valence trounce, the virtually stable electron configuration. The periodic table is arranged in columns and rows based on the number of electrons and where these electrons are located, providing a tool to sympathize how electrons are distributed in the outer vanquish of an cantlet. As shown in, the group 18 atoms helium (He), neon (Ne), and argon (Ar) all have filled outer electron shells, making it unnecessary for them to gain or lose electrons to attain stability; they are highly stable as single atoms. Their non-reactivity has resulted in their being named the inert gases (or noble gases). In comparison, the group 1 elements, including hydrogen (H), lithium (Li), and sodium (Na), all have one electron in their outermost shells. This ways that they tin achieve a stable configuration and a filled outer shell past donating or losing an electron. Equally a result of losing a negatively-charged electron, they become positively-charged ions. When an atom loses an electron to become a positively-charged ion, this is indicated by a plus sign afterward the element symbol; for case, Na+. Group 17 elements, including fluorine and chlorine, take seven electrons in their outermost shells; they tend to fill this beat by gaining an electron from other atoms, making them negatively-charged ions. When an cantlet gains an electron to become a negatively-charged ion this is indicated by a minus sign after the element symbol; for case, F-. Thus, the columns of the periodic table represent the potential shared land of these elements' outer electron shells that is responsible for their similar chemical characteristics.
Electron Orbitals
Electron orbitals are three-dimensional representations of the space in which an electron is probable to be found.
Learning Objectives
Distinguish between electron orbitals in the Bohr model versus the quantum mechanical orbitals
Key Takeaways
Key Points
- The Bohr model of the cantlet does non accurately reflect how electrons are spatially distributed around the nucleus equally they do non circle the nucleus similar the earth orbits the sun.
- The electron orbitals are the result of mathematical equations from quantum mechanics known as wave functions and can predict inside a sure level of probability where an electron might be at whatever given time.
- The number and blazon of orbitals increases with increasing atomic number, filling in various electron shells.
- The area where an electron is most likely to be found is called its orbital.
Fundamental Terms
- electron shell: The commonage states of all electrons in an cantlet having the aforementioned principal quantum number (visualized as an orbit in which the electrons move).
- orbital: A specification of the energy and probability density of an electron at any point in an atom or molecule.
Although useful to explain the reactivity and chemical bonding of certain elements, the Bohr model of the atom does not accurately reflect how electrons are spatially distributed surrounding the nucleus. They practise not circle the nucleus like the earth orbits the sun, merely are rather found in electron orbitals. These relatively circuitous shapes upshot from the fact that electrons behave not just similar particles, but as well similar waves. Mathematical equations from quantum mechanics known every bit moving ridge functions can predict within a certain level of probability where an electron might be at whatsoever given time. The expanse where an electron is nigh likely to be plant is called its orbital.
Starting time Electron Shell
The closest orbital to the nucleus, called the 1s orbital, tin hold upwardly to two electrons. This orbital is equivalent to the innermost electron shell of the Bohr model of the cantlet. It is chosen the 1s orbital because it is spherical around the nucleus. The 1s orbital is always filled before whatsoever other orbital. Hydrogen has one electron; therefore, it has simply one spot within the 1s orbital occupied. This is designated equally 1s1, where the superscripted 1 refers to the one electron within the 1s orbital. Helium has ii electrons; therefore, it tin can completely make full the 1s orbital with its two electrons. This is designated equally 1stwo, referring to the two electrons of helium in the 1s orbital. On the periodic table, hydrogen and helium are the only two elements in the first row (period); this is considering they are the sole elements to have electrons only in their first shell, the 1s orbital.
Second Electron Shell
The 2nd electron trounce may comprise eight electrons. This shell contains another spherical s orbital and iii "dumbbell" shaped p orbitals, each of which can hold two electrons. Later the 1s orbital is filled, the 2nd electron beat out is filled, kickoff filling its 2s orbital and then its 3 p orbitals. When filling the p orbitals, each takes a unmarried electron; once each p orbital has an electron, a second may be added. Lithium (Li) contains three electrons that occupy the first and second shells. 2 electrons fill the 1s orbital, and the 3rd electron then fills the 2s orbital. Its electron configuration is 1stwo2s1. Neon (Ne), on the other paw, has a total of ten electrons: two are in its innermost 1s orbital, and eight fill up its 2d trounce (ii each in the 2s and 3 p orbitals). Thus, information technology is an inert gas and energetically stable: it rarely forms a chemical bond with other atoms.

Diagram of the S and P orbitals: The s subshells are shaped like spheres. Both the 1n and 2n principal shells have an s orbital, merely the size of the sphere is larger in the 2n orbital. Each sphere is a unmarried orbital. p subshells are made upwardly of iii dumbbell-shaped orbitals. Principal shell 2n has a p subshell, but trounce 1 does not.
Third Electron Trounce
Larger elements have additional orbitals, making up the tertiary electron shell. Subshells d and f have more complex shapes and contain five and 7 orbitals, respectively. Principal shell 3n has s, p, and d subshells and can hold 18 electrons. Principal shell 4n has due south, p, d, and f orbitals and can agree 32 electrons. Moving abroad from the nucleus, the number of electrons and orbitals institute in the energy levels increases. Progressing from one atom to the next in the periodic table, the electron structure can exist worked out past plumbing equipment an extra electron into the adjacent bachelor orbital. While the concepts of electron shells and orbitals are closely related, orbitals provide a more than authentic depiction of the electron configuration of an cantlet considering the orbital model specifies the different shapes and special orientations of all the places that electrons may occupy.
Chemical Reactions and Molecules
Chemical reactions occur when 2 or more than atoms bond together to course molecules or when bonded atoms are broken autonomously.
Learning Objectives
Describe the properties of chemical reactions and compounds
Key Takeaways
Key Points
- Atoms form chemical bonds with other atoms thereby obtaining the electrons they demand to attain a stable electron configuration.
- The substances used in the beginning of a chemical reaction are called the reactants and the substances found at the end of the reaction are known as the products.
- Some reactions are reversible and will reach a relative balance between reactants and products: a country called equilibrium.
- An arrow is typically drawn betwixt the reactants and products to indicate the direction of the chemic reaction.
Central Terms
- reactant: Any of the participants present at the first of a chemical reaction.
- molecule: The smallest particle of a specific chemical compound that retains the chemical backdrop of that compound; ii or more than atoms held together past chemic bonds.
- reaction: A transformation in which ane or more than substances is converted into another by combination or decomposition
Chemical Reactions and Molecules
According to the octet rule, elements are most stable when their outermost shell is filled with electrons. This is because it is energetically favorable for atoms to be in that configuration. However, since not all elements take plenty electrons to fill their outermost shells, atoms form chemic bonds with other atoms, which helps them obtain the electrons they need to accomplish a stable electron configuration. When 2 or more atoms chemically bond with each other, the resultant chemical structure is a molecule. The familiar water molecule, H2O, consists of two hydrogen atoms and one oxygen atom, which bond together to form h2o. Atoms can grade molecules by altruistic, accepting, or sharing electrons to fill their outer shells.

Atoms bond to form molecules: Ii or more atoms may bond with each other to course a molecule. When two hydrogens and an oxygen share electrons via covalent bonds, a water molecule is formed.
Chemic reactions occur when two or more atoms bond together to grade molecules or when bonded atoms are cleaved apart. The substances used in the beginning of a chemic reaction are called the reactants (commonly found on the left side of a chemical equation), and the substances found at the end of the reaction are known as the products (usually constitute on the right side of a chemical equation). An pointer is typically drawn between the reactants and products to indicate the direction of the chemical reaction. For the cosmos of the h2o molecule shown to a higher place, the chemical equation would exist:
[latex]two\text{H}_2+\text{O}_2\rightarrow2\text{H}_2\text{O}[/latex]
An example of a unproblematic chemical reaction is the breaking downwards of hydrogen peroxide molecules, each of which consists of two hydrogen atoms bonded to two oxygen atoms (H2Otwo). The reactant hydrogen peroxide is broken downward into h2o (HtwoO), and oxygen, which consists of 2 bonded oxygen atoms (Otwo). In the equation beneath, the reaction includes ii hydrogen peroxide molecules and two h2o molecules. This is an example of a balanced chemical equation, wherein the number of atoms of each element is the same on each side of the equation. Co-ordinate to the constabulary of conservation of thing, the number of atoms before and after a chemic reaction should exist equal, such that no atoms are, under normal circumstances, created or destroyed.
[latex]2\text{H}_2\text{O}_2 \rightarrow two\text{H}_2\text{O} + \text{O}_2[/latex]
Even though all of the reactants and products of this reaction are molecules (each atom remains bonded to at least one other cantlet), in this reaction just hydrogen peroxide and water are representative of a subclass of molecules known as compounds: they contain atoms of more 1 type of element. Molecular oxygen, on the other hand, consists of two doubly bonded oxygen atoms and is not classified as a compound simply as an chemical element.
Irreversible and Reversible Reactions
Some chemical reactions, such as the one shown to a higher place, tin proceed in one direction until the reactants are all used up. The equations that describe these reactions incorporate a unidirectional arrow and are irreversible. Reversible reactions are those that tin can go in either direction. In reversible reactions, reactants are turned into products, but when the concentration of production goes beyond a certain threshold, some of these products will be converted back into reactants; at this point, the designations of products and reactants are reversed. This dorsum and forth continues until a certain relative balance between reactants and products occurs: a state called equilibrium. These situations of reversible reactions are often denoted past a chemic equation with a double headed arrow pointing towards both the reactants and products.
For example, in human claret, excess hydrogen ions (H+) bind to bicarbonate ions (HCO3 –) forming an equilibrium state with carbonic acid (HtwoCOthree). If carbonic acrid were added to this system, some of it would exist converted to bicarbonate and hydrogen ions.
In biological reactions, nonetheless, equilibrium is rarely obtained because the concentrations of the reactants or products or both are constantly changing, often with a product of one reaction beingness a reactant for another. To return to the instance of backlog hydrogen ions in the claret, the formation of carbonic acid will be the major direction of the reaction. However, the carbonic acid can also leave the body as carbon dioxide gas (via exhalation) instead of beingness converted back to bicarbonate ion, thus driving the reaction to the correct past the chemical law known as law of mass action. These reactions are important for maintaining the homeostasis of our claret.
Ions and Ionic Bonds
Ionic bonds are attractions betwixt oppositely charged atoms or groups of atoms where electrons are donated and accepted.
Learning Objectives
Predict whether a given chemical element will more likely form a cation or an anion
Key Takeaways
Key Points
- Ions form from elements when they proceeds or lose an electron causing the number of protons to be unequal to the number of electrons, resulting in a net charge.
- If there are more electrons than protons (from an element gaining ane or more electrons), the ion is negatively charged and called an anion.
- If there are more than protons than electrons (via loss of electrons), the ion is positively charged and is called a cation.
- Ionic bonds result from the interaction between a positively charged cation and a negatively charged anion.
Cardinal Terms
- ion: An atom, or group of atoms, bearing an electrical charge, such equally the sodium and chlorine atoms in a salt solution.
- ionic bail: A strong chemic bond caused past the electrostatic attraction between two oppositely charged ions.
Ions and Ionic Bonds
Some atoms are more stable when they gain or lose an electron (or maybe ii) and form ions. This results in a full outermost electron beat out and makes them energetically more stable. At present, considering the number of electrons does non equal the number of protons, each ion has a net accuse. Cations are positive ions that are formed by losing electrons (every bit the number of protons is now greater than the number of electrons). Negative ions are formed by gaining electrons and are called anions (wherein in that location are more electrons than protons in a molecule ). Anions are designated by their elemental name being altered to end in "-ide". For example, the anion of chlorine is chosen chloride, and the anion of sulfur is chosen sulfide.
This movement of electrons from one chemical element to some other is referred to as electron transfer. As illustrated, sodium (Na) only has one electron in its outer electron shell. It takes less free energy for sodium to donate that ane electron than it does to take seven more electrons to make full the outer crush. When sodium loses an electron, it will have eleven protons, 11 neutrons, and just x electrons. This leaves it with an overall charge of +1 since there are now more protons than electrons. It is now referred to as a sodium ion. Chlorine (Cl) in its lowest energy country (called the ground state) has 7 electrons in its outer trounce. Once more, it is more energy efficient for chlorine to gain one electron than to lose seven. Therefore, it tends to gain an electron to create an ion with 17 protons, 17 neutrons, and 18 electrons. This gives information technology a net charge of -one since at that place are now more electrons than protons. Information technology is now referred to equally a chloride ion. In this example, sodium will donate its one electron to empty its trounce, and chlorine will accept that electron to make full its shell. Both ions at present satisfy the octet rule and have complete outer shells. These transactions can normally only take identify simultaneously; in order for a sodium atom to lose an electron, it must be in the presence of a suitable recipient like a chlorine atom.

Electron Transfer Between Na and Cl: In the germination of an ionic compound, metals lose electrons and nonmetals proceeds electrons to achieve an octet. In this example, sodium loses one electron to empty its shell and chlorine accepts that electron to make full its shell.
Ionic bonds are formed between ions with opposite charges. For instance, positively charged sodium ions and negatively charged chloride ions bond together to form sodium chloride, or table salt, a crystalline molecule with zero net charge. The attractive strength holding the two atoms together is called the electromagnetic force and is responsible for the allure between oppositely charged ions.
Certain salts are referred to in physiology as electrolytes (including sodium, potassium, and calcium). Electrolytes are ions necessary for nerve impulse conduction, muscle contractions, and h2o residual. Many sports drinks and dietary supplements provide these ions to supercede those lost from the body via sweating during practise.
Covalent Bonds and Other Bonds and Interactions
Covalent bonds result from a sharing of electrons between ii atoms and concur most biomolecules together.
Learning Objectives
Compare the relative forcefulness of different types of bonding interactions
Cardinal Takeaways
Key Points
- A polar covalent bond arises when ii atoms of different electronegativity share ii electrons unequally.
- A non-polar covalent bond is i in which the electrons are shared equally between 2 atoms.
- Hydrogen bonds and Van Der Waals are responsible for the folding of proteins, the binding of ligands to proteins, and many other processes betwixt molecules.
Fundamental Terms
- hydrogen bail: A weak bond in which a hydrogen atom in one molecule is attracted to an electronegative atom (usually nitrogen or oxygen) in the aforementioned or different molecule.
- covalent bond: A type of chemical bond where two atoms are connected to each other by the sharing of two or more than electrons.
- dipole: Any object (such as a magnet, polar molecule or antenna), that is oppositely charged at 2 points (or poles).
Covalent Bonds and Other Bonds and Interactions
The octet dominion can be satisfied by the sharing of electrons between atoms to form covalent bonds. These bonds are stronger and much more common than are ionic bonds in the molecules of living organisms. Covalent bonds are usually institute in carbon-based organic molecules, such as Dna and proteins. Covalent bonds are also institute in inorganic molecules such as H2O, COtwo, and O2. One, 2, or three pairs of electrons may exist shared between 2 atoms, making single, double, and triple bonds, respectively. The more covalent bonds between two atoms, the stronger their connection. Thus, triple bonds are the strongest.
The strength of dissimilar levels of covalent bonding is one of the main reasons living organisms have a difficult fourth dimension in acquiring nitrogen for use in constructing nitrogenous molecules, fifty-fifty though molecular nitrogen, N2, is the most abundant gas in the atmosphere. Molecular nitrogen consists of two nitrogen atoms triple bonded to each other. The resulting strong triple bail makes information technology difficult for living systems to suspension apart this nitrogen in order to use it every bit constituents of biomolecules, such as proteins, DNA, and RNA.
The formation of water molecules is an example of covalent bonding. The hydrogen and oxygen atoms that combine to form water molecules are leap together by covalent bonds. The electron from the hydrogen splits its time between the incomplete outer vanquish of the hydrogen atom and the incomplete outer shell of the oxygen cantlet. In return, the oxygen atom shares one of its electrons with the hydrogen atom, creating a two-electron unmarried covalent bond. To completely fill the outer trounce of oxygen, which has vi electrons in its outer shell, 2 electrons (one from each hydrogen atom) are needed. Each hydrogen atom needs simply a unmarried electron to fill its outer crush, hence the well-known formula H2O. The electrons that are shared between the two elements fill the outer shell of each, making both elements more than stable.
Polar Covalent Bonds
There are two types of covalent bonds: polar and nonpolar. In a polar covalent bond, the electrons are unequally shared past the atoms because they are more attracted to one nucleus than the other. The relative allure of an atom to an electron is known as its electronegativity: atoms that are more than attracted to an electron are considered to be more electronegative. Because of the unequal distribution of electrons between the atoms of different elements, a slightly positive (δ+) or slightly negative (δ-) charge develops. This partial charge is known equally a dipole; this is an important property of water and accounts for many of its characteristics. The dipole in h2o occurs because oxygen has a higher electronegativity than hydrogen, which means that the shared electrons spend more time in the vicinity of the oxygen nucleus than they do almost the nucleus of the hydrogen atoms.
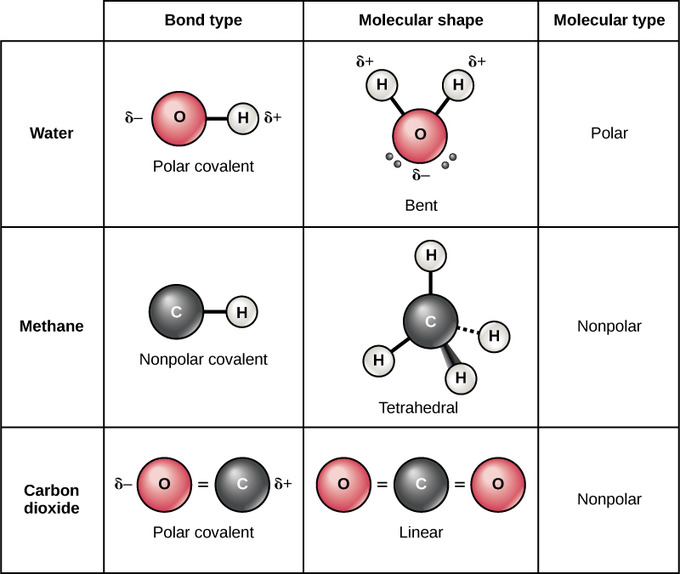
Polar and Nonpolar Covalent Bonds: Whether a molecule is polar or nonpolar depends both on bond blazon and molecular shape. Both water and carbon dioxide take polar covalent bonds, but carbon dioxide is linear, so the fractional charges on the molecule cancel each other out.
Nonpolar Covalent Bonds
Nonpolar covalent bonds form between ii atoms of the same element or between dissimilar elements that share electrons equally. For example, molecular oxygen (O2) is nonpolar because the electrons will be equally distributed between the ii oxygen atoms. The iv bonds of methane are also considered to be nonpolar because the electronegativies of carbon and hydrogen are nearly identical.
Hydrogen Bonds and Van Der Waals Interactions
Not all bonds are ionic or covalent; weaker bonds can besides class between molecules. 2 types of weak bonds that oft occur are hydrogen bonds and van der Waals interactions. Without these two types of bonds, life as we know it would not exist.
Hydrogen bonds provide many of the critical, life-sustaining properties of water and also stabilize the structures of proteins and Dna, the edifice block of cells. When polar covalent bonds containing hydrogen are formed, the hydrogen atom in that bond has a slightly positive accuse (δ+) considering the shared electrons are pulled more strongly toward the other element and away from the hydrogen cantlet. Because the hydrogen has a slightly positive charge, information technology's attracted to neighboring negative charges. The weak interaction betwixt the δ+ charge of a hydrogen atom from i molecule and the δ- charge of a more electronegative atom is called a hydrogen bail. Private hydrogen bonds are weak and easily cleaved; nonetheless, they occur in very large numbers in h2o and in organic polymers, and the additive strength can be very strong. For example, hydrogen bonds are responsible for zipping together the DNA double helix.

Adenosine Triphosphate, ATP: Adenosine Triphosphate, or ATP, is the about commonly used cofactor in nature. Its biosynthesis involves the fixation of nitrogen to provide feedstocks that eventually produce the carbon-nitrogen bonds information technology contains.
Like hydrogen bonds, van der Waals interactions are weak interactions betwixt molecules. Van der Waals attractions tin can occur betwixt whatsoever 2 or more molecules and are dependent on slight fluctuations of the electron densities, which can atomic number 82 to slight temporary dipoles effectually a molecule. For these attractions to happen, the molecules need to be very close to one another. These bonds, forth with hydrogen bonds, assist form the three-dimensional structures of the proteins in our cells that are required for their proper function.
Hydrogen Bonding and Van der Waals Forces
Hydrogen bonds and van der Waals interactions are two types of weak bonds that are necessary to the basic building blocks of life.
Learning Objectives
Draw how hydrogen bonds and van der Waals interactions occur
Cardinal Takeaways
Fundamental Points
- Hydrogen bonds provide many of the disquisitional, life-sustaining properties of water and too stabilize the structures of proteins and DNA, the building block of cells.
- Hydrogen bonds occur in inorganic molecules, such as water, and organic molecules, such as Dna and proteins.
- Van der Waals attractions can occur betwixt whatsoever 2 or more molecules and are dependent on slight fluctuations of the electron densities.
- While hydrogen bonds and van der Waals interactions are weak individually, they are strong combined in vast numbers.
Key Terms
- van der Waals interactions: A weak force of attraction betwixt electrically neutral molecules that collide with or pass very close to each other. The van der Waals strength is caused by temporary attractions between electron-rich regions of ane molecule and electron-poor regions of another.
- electronegativity: The tendency of an cantlet or molecule to draw electrons towards itself, course dipoles, and thus form bonds.
- hydrogen bail: The attraction between a partially positively-charged hydrogen cantlet attached to a highly electronegative atom (such equally nitrogen, oxygen, or fluorine) and some other nearby electronegative atom.
Ionic and covalent bonds betwixt elements require energy to interruption. Ionic bonds are not as strong as covalent, which determines their behavior in biological systems. Withal, non all bonds are ionic or covalent bonds. Weaker bonds can also form between molecules. Two weak bonds that occur frequently are hydrogen bonds and van der Waals interactions.

Hydrogen bonds between h2o molecules: The slightly negative oxygen side of the water molecule and the slightly positive hydrogen side of the water molecule are attracted to each other and grade a hydrogen bond.
Hydrogen Bonding
Hydrogen bonds provide many of the critical, life-sustaining properties of water and likewise stabilize the structures of proteins and DNA, the edifice block of cells. When polar covalent bonds containing hydrogen form, the hydrogen in that bond has a slightly positive charge because hydrogen's one electron is pulled more than strongly toward the other chemical element and away from the hydrogen. Considering the hydrogen is slightly positive, it will be attracted to neighboring negative charges. When this happens, an interaction occurs between the δ +of the hydrogen from one molecule and the δ– charge on the more electronegative atoms of some other molecule, commonly oxygen or nitrogen, or within the same molecule. This interaction is called a hydrogen bail. This type of bond is common and occurs regularly between water molecules. Individual hydrogen bonds are weak and easily cleaved; however, they occur in very large numbers in water and in organic polymers, creating a major force in combination. Hydrogen bonds are also responsible for zipping together the DNA double helix.
Applications for Hydrogen Bonds
Hydrogen bonds occur in inorganic molecules, such equally h2o, and organic molecules, such equally Dna and proteins. The two complementary strands of Dna are held together past hydrogen bonds between complementary nucleotides (A&T, C&Grand). Hydrogen bonding in water contributes to its unique properties, including its high boiling bespeak (100 °C) and surface tension.

Water droplets on a leaf: The hydrogen bonds formed between water molecules in water droplets are stronger than the other intermolecular forces between the water molecules and the leaf, contributing to high surface tension and distinct h2o aerosol.
In biology, intramolecular hydrogen bonding is partly responsible for the secondary, 3rd, and fourth structures of proteins and nucleic acids. The hydrogen bonds help the proteins and nucleic acids course and maintain specific shapes.
Van der Waals Interactions
Like hydrogen bonds, van der Waals interactions are weak attractions or interactions betwixt molecules. Van der Waals attractions can occur between whatsoever two or more molecules and are dependent on slight fluctuations of the electron densities, which are not always symmetrical around an cantlet. For these attractions to happen, the molecules demand to be very shut to ane another. These bonds—along with ionic, covalent, and hydrogen bonds—contribute to the three-dimensional structure of proteins that is necessary for their proper office.
Source: https://courses.lumenlearning.com/boundless-biology/chapter/atoms-isotopes-ions-and-molecules/
0 Response to "for an atom to achieve maximum stability and become chemically inert, what must occur?"
Post a Comment